How to move to post-Brexit pharmacovigilance effortlessly and efficiently
November 27, 2020
In 2021 the United Kingdom is moving to its post-Brexit life with new processes, procedures, and laws everywhere, including clinical trials and pharmacovigilance. This piece we dedicate to a simple question – how to move to post-Brexit pharmacovigilance effortlessly and efficiently.
All information in this article is based on Updated guidance on pharmacovigilance procedures published by Medicines & Healthcare products Regulatory Agency, last updated 27 October 2020.
Let’s dive into updated submissions data:
When: From 1 January 2021
Who: Marketing Authorisation Holders (MAH) of medicines authorised in Great Britain
Where to submit: to the MHRA
What to submit: all pharmacovigilance data, including
- UK and non-UK Individual Case Safety Reports (ICSRs)
- Periodic Safety Update Reports (PSURs)
- Risk Management Plans (RMPs)
- Post-Authorisation Safety Studies (PASS) protocols and final study reports
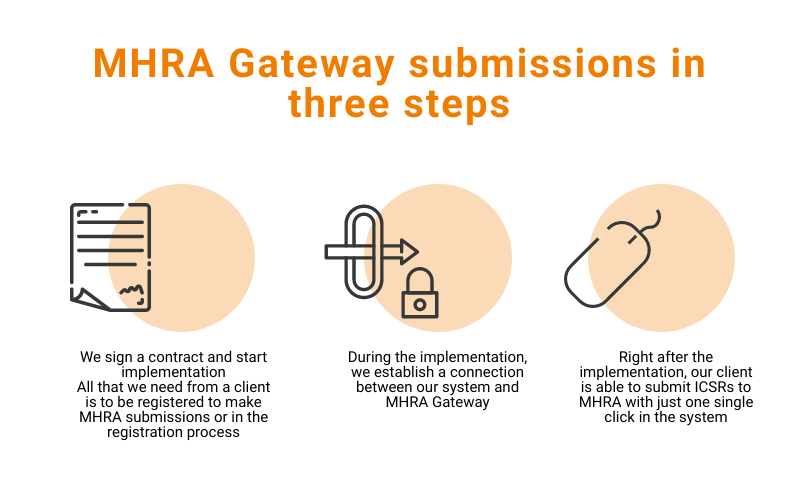
Flex Databases Pharmacovigilance is here to help you with the new MHRA Gateway submissions of ICSRs.
Submitting Individual Case Safety Reports
As the MHRA states, “We will require submission of all UK (including NI) ICSRs (serious and non-serious) and serious ICSRs from other countries via the new MHRA Gateway or/ICSR Submissions portal which have been developed.”
The MHRA recommends registering for submissions “as early as possible”.
Move to compliant PV system within a month before regulation changes? We say let’s do it!
To start moving to post-Brexit pharmacovigilance, request a quote or learn more request a demo via the button on top of the page, or send us an e-mail to bd@flexdatabases.com!



