




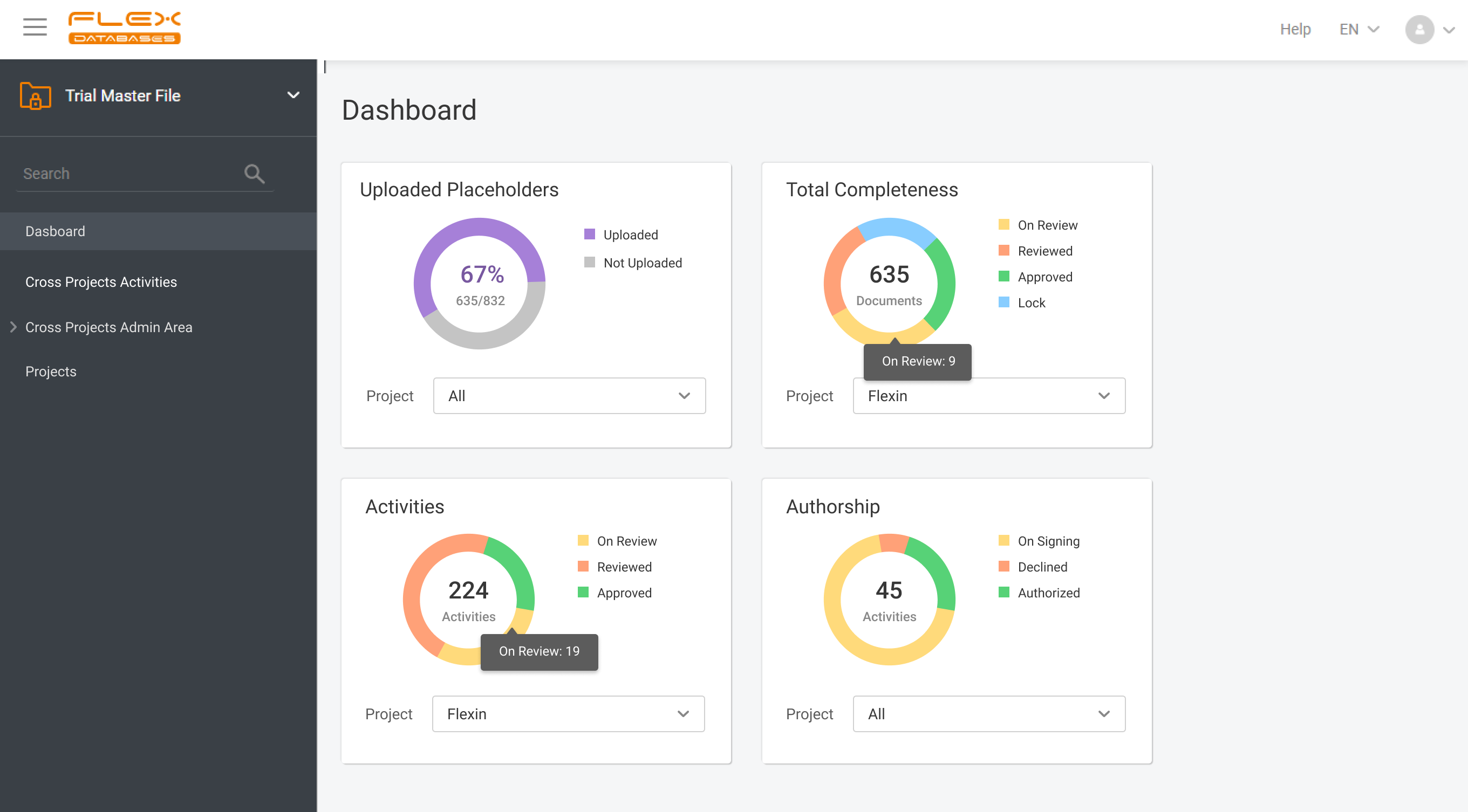
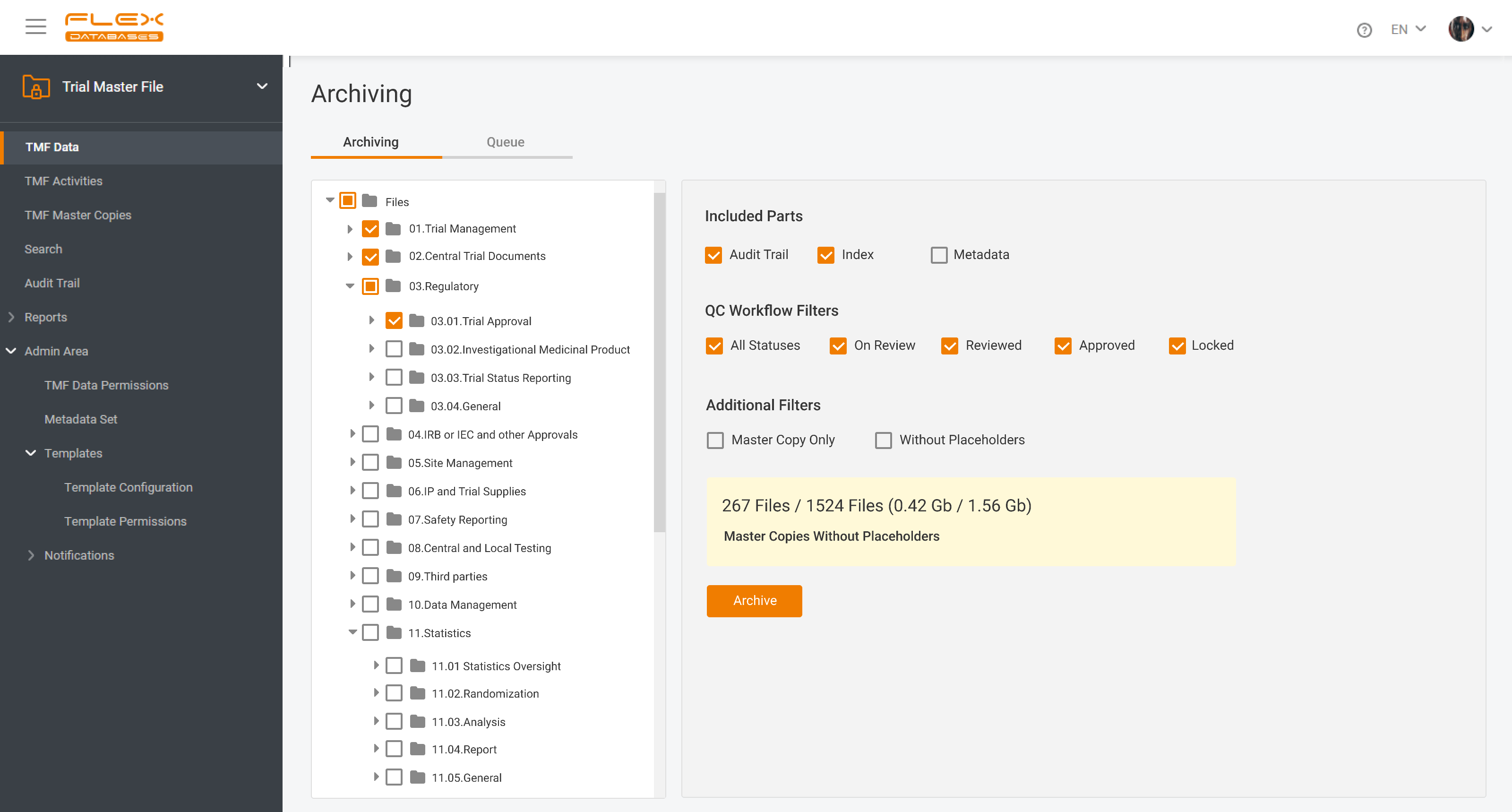
eTMF – complete 24/7 visibility into all your trial documentation





Drop us a line and we be happy to answer all your questions
Clients
Daily users
Documents
Clinical trials
Countries
Blinded studies are critical for maintaining the integrity of clinical trials. But while the concept is straightforward, the operational reality is often anything but. For many clinical teams, managing blinding means dealing with: This creates a constant burden: Even with strict processes in place, a single misconfiguration can compromise the entire study. TMF Blinding in […]
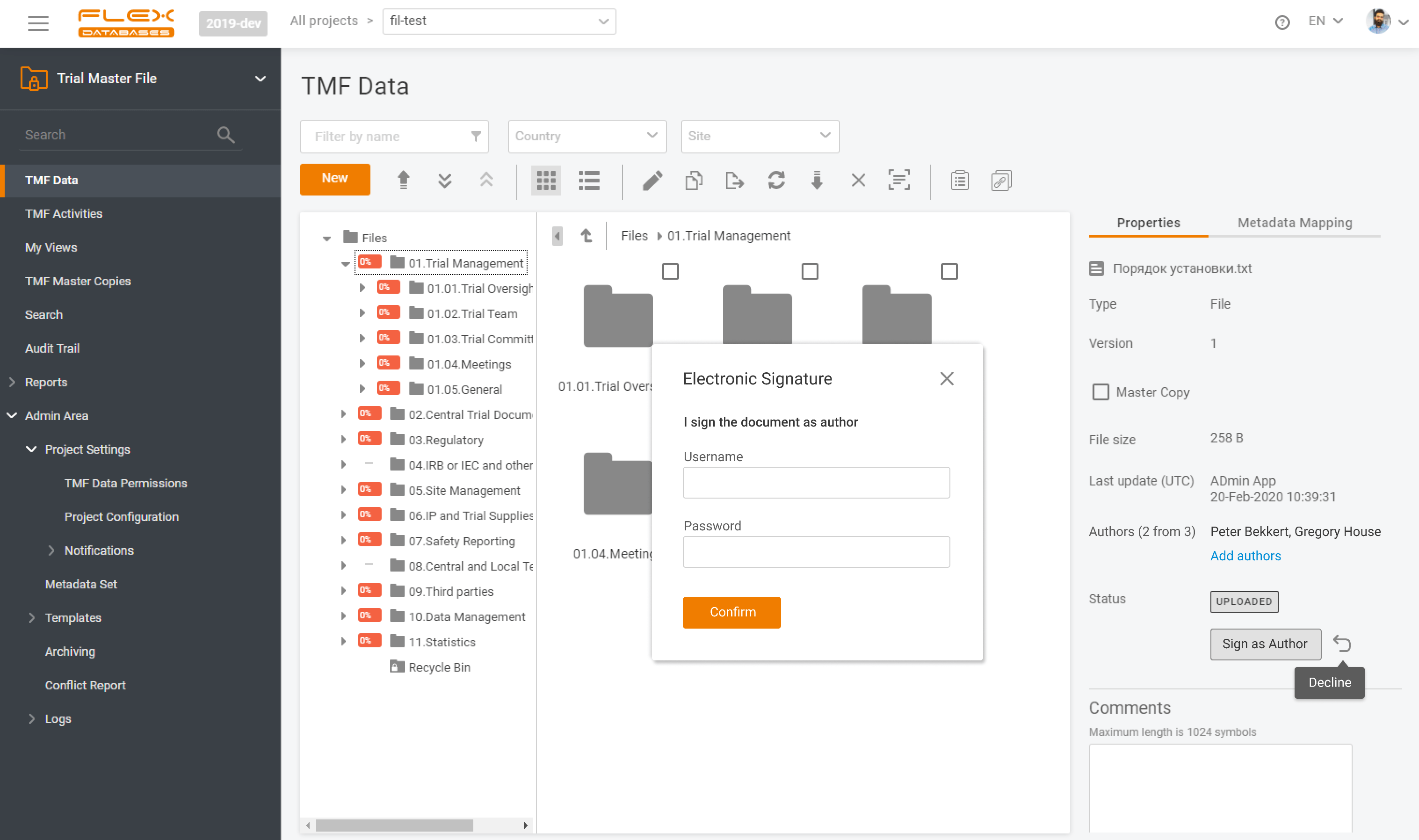
To learn more about the reasons behind this decision, we asked Tom Apelt, IT Manager at Immutep GmbH, to share his perspective on selecting Flex Databases and the experience of implementing the eTMF system. Before implementing Flex Databases eTMF, what were the main challenges you experienced in TMF management? Prior to implementing Flex Databases eTMF, […]
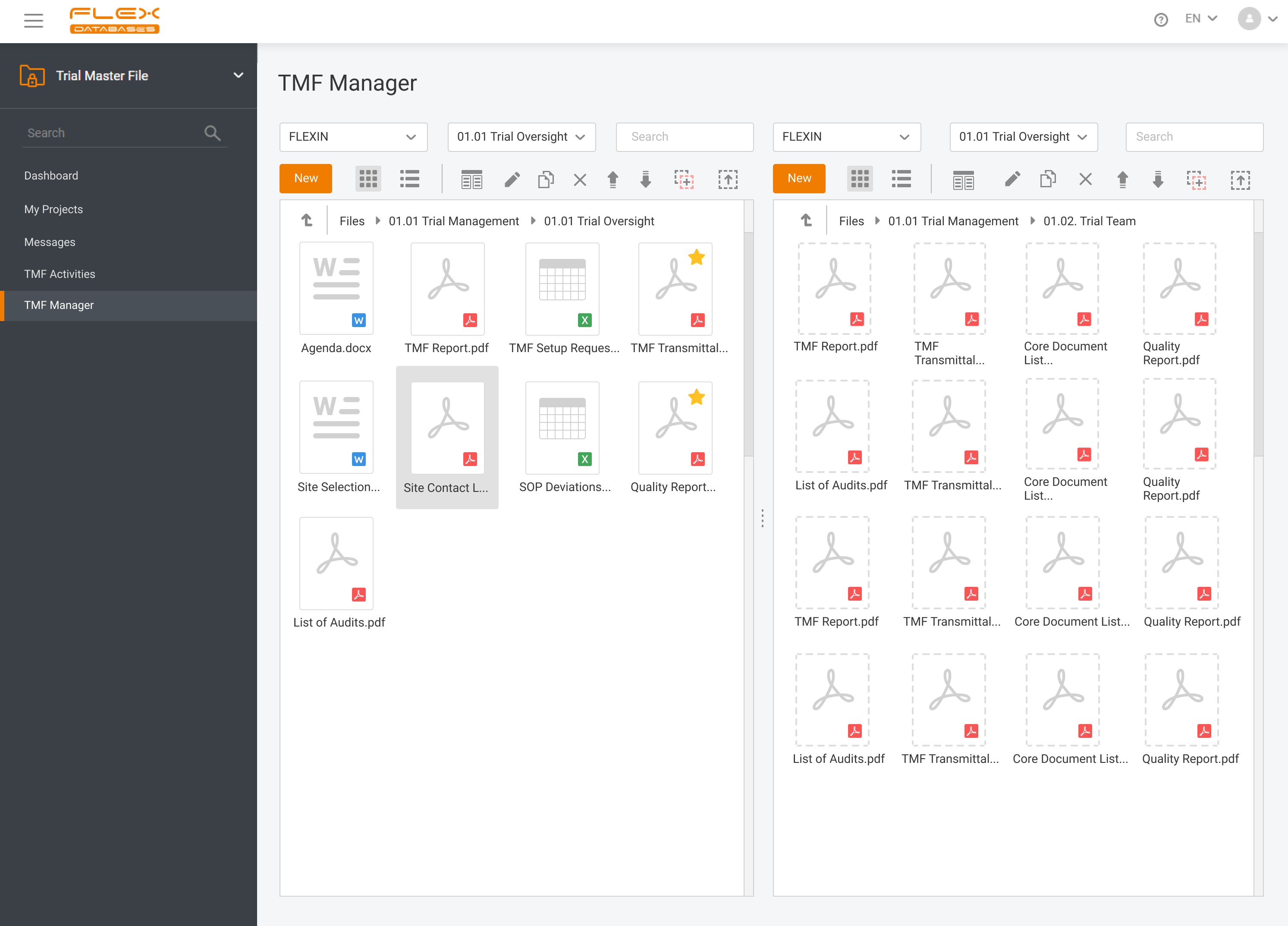
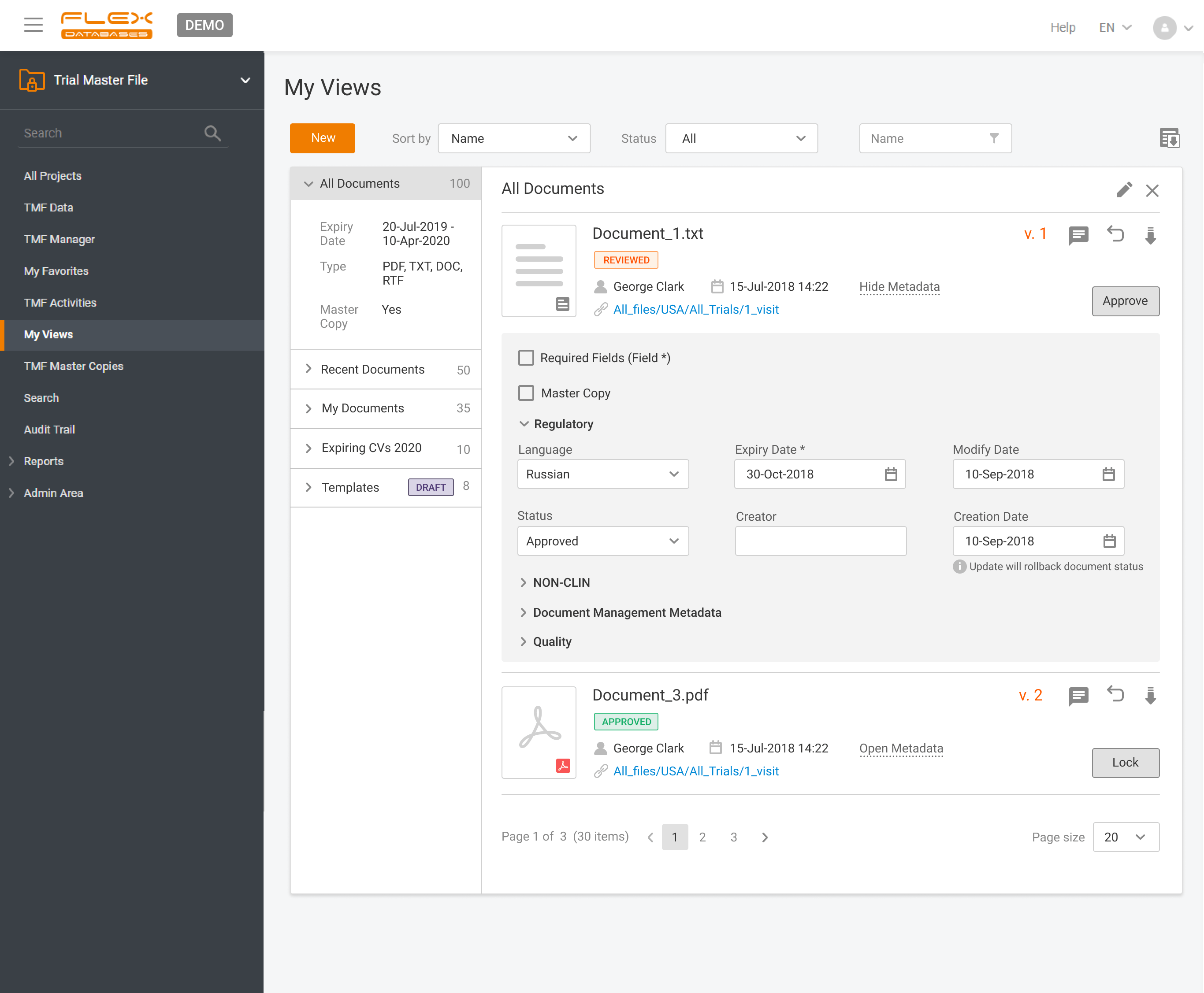
Flex Databases Solves One of the Biggest Document Management Headaches Clinical teams have long struggled with one persistent problem: managing TMF documents across multiple systems, versions, and stakeholders. Switching between platforms, downloading files, losing track of versions, and waiting for colleagues to finish editing slows down operations and increases compliance risks. With this update, Flex […]
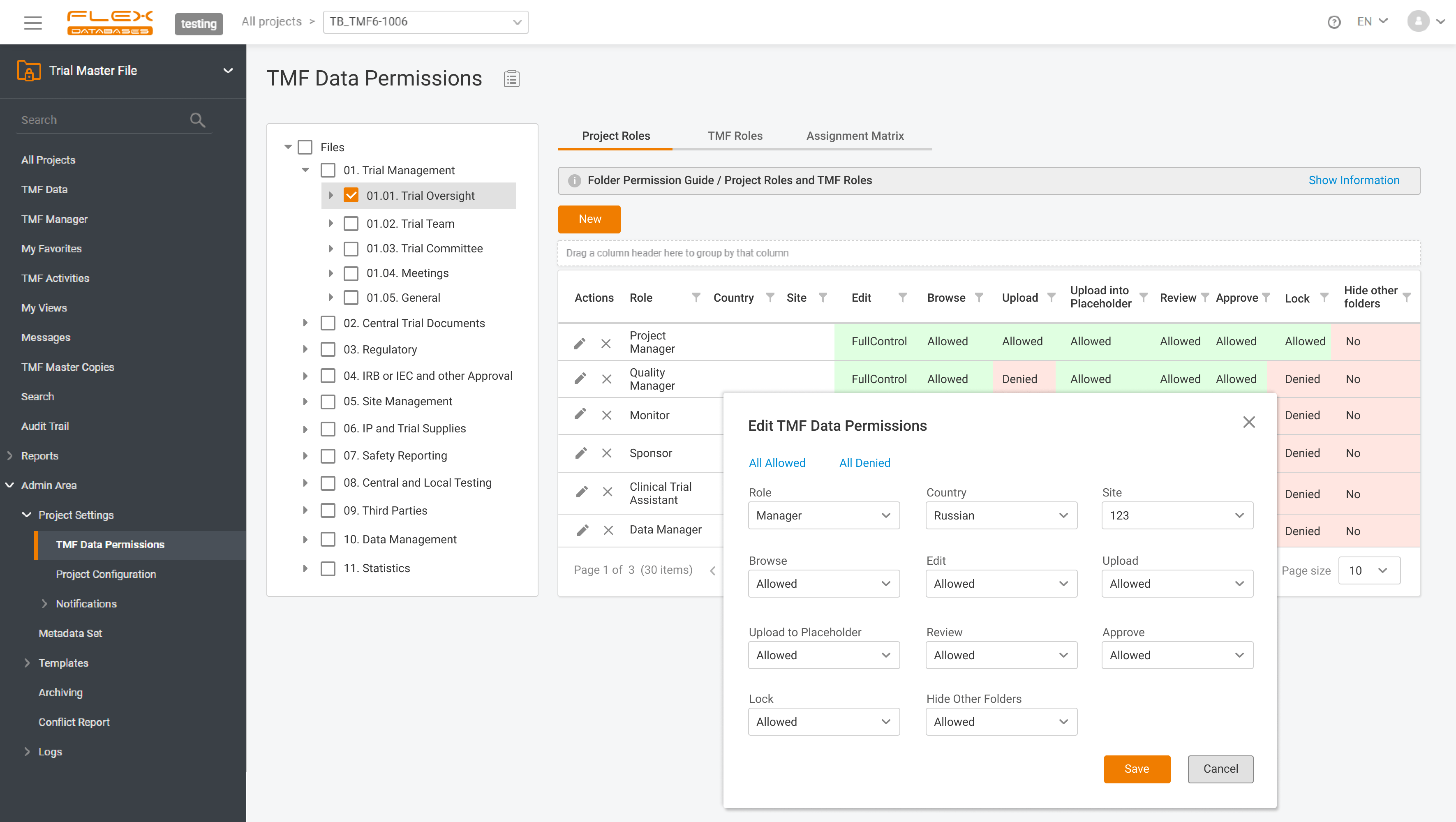
📂 Last-minute TMF quality reviews cause stress, missed documents, and higher audit risks.With Flex Databases, you avoid the scramble – reviews are structured, transparent, and always under your control. With Flex Databases, you get:✅ Control over the process✅ Full transparency and traceability✅ Less stress before audits✅ Clear outcomes in one report 🎥 See how it […]
Get in touch to discuss compliance, implementation, demos, pricing
We are here for all of your questions! Tell us more about yourself and we will organize a tailored live demo to show how you can power up your clinical trials processes with Flex Databases.