Bionical Emas is the only CRO to combine Clinical Development, Early Access Programs and Clinical Trial Supply, to deliver a unique, seamless approach supporting our clients to bring new medicines to patients faster.
Blog Flex Databases

United States-based Tranquil Clinical Research recently signed Flex Databases as a software provider for Clinical Trial Management System and Pharmacovigilance management solution.

This June Assign Data Management and Biostatistics – Assign DMB chose Flex Databases as a provider for CTMS, eTMF, and Pharmacovigilance management systems.

Sourcia, a full-service European CRO, has recently signed a contract for electronic Trial Master File implementation with Flex Databases.
Why customers value Flex Databases
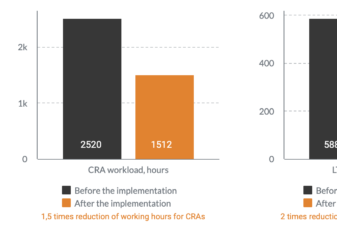
How Novo Nordisk sped up payment process and cut costs with Flex Databases CTMS
A2 Healthcare Taiwan Corporation (A2TW) has recently adopted Flex Databases CTMS and TMF solutions.
At DIA 2019 Yvonne Lungershausen, CEO Avance Clinical, and Evgenia Mikhalchuk, Director, Business Development & Marketing Flex Databases discussed with PharmaVoice how Avance Clinical performed vendor selection for CTMS and TMF and why they chose Flex Databases. Watch the full interview (5 min.) to know more!

Last week Timur Galimov, our Chief Technology Officer, and Olga Sokolova, System Analysis Director, conducted a 2-day training for our client Novo Nordisk, a global healthcare company with more than 90 years history of innovation and leadership in diabetes care. Flex Databases team walked Novo Nordisk team through the system, demonstrated functionality peculiarities. The system […]