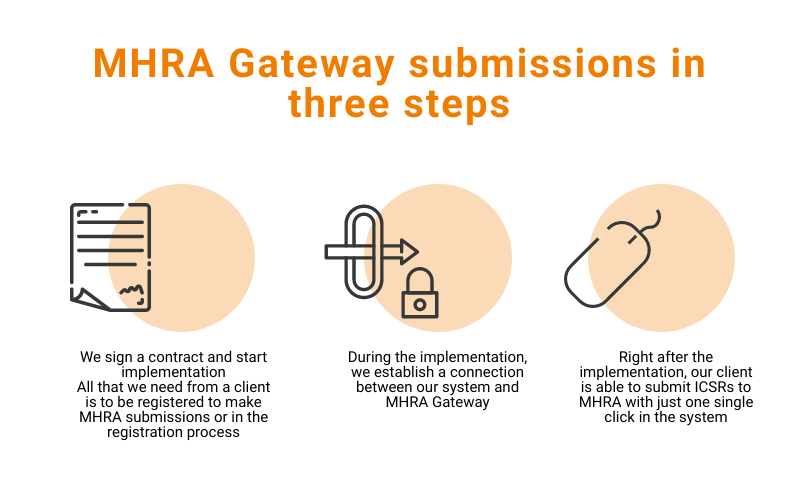
Blog Flex Databases
In 2021 the United Kingdom is moving to its post-Brexit life with new processes, procedures, and laws everywhere, including clinical trials and pharmacovigilance. This piece we dedicate to a simple question – how to move to post-Brexit pharmacovigilance effortlessly and efficiently. All information in this article is based on Updated guidance on pharmacovigilance procedures published by Medicines […]
United States-based Tranquil Clinical Research recently signed Flex Databases as a software provider for Clinical Trial Management System and Pharmacovigilance management solution. Tranquil’s ultimate goal is excellence in the clinical trial process and bringing trustworthy products to patients. Flex Databases CTMS and Pharmacovigilance system are well-known as unified yet flexible eClinical platform for full-cycle clinical trial management. Baseline implementation takes […]
Flex Databases Signal Management tool in Pharmacovigilance module utilizes modern qualitative and quantitative (statistical, machine learning, neural network) methods to detect signals more precisely. And, from now on, it is provided for free for all Pharmacovigilance module buyers. Request a demo via the button on top of the page to know more!
The drug regulatory authority of China – National Medical Product Administration (NMPA, formerly CFDA) has recently implemented a possibility to report Adverse Drug Reactions (ADR) through an XML-file submission. Flex Databases Pharmacovigilance system users are already able to do so. There are two general ways of reporting to the regulator – manual submissions through the website and […]