Five ways in which clinical study can benefit from having CTMS in place
April 21, 2023
“Why did you decide to implement CTMS?” is one of the first questions our CTMS clients get during a presale. We’ve collected top-5 answers for you to check and see if something hits close to home, and it’s time to start thinking about your own CTMS implementation.
Here they are:
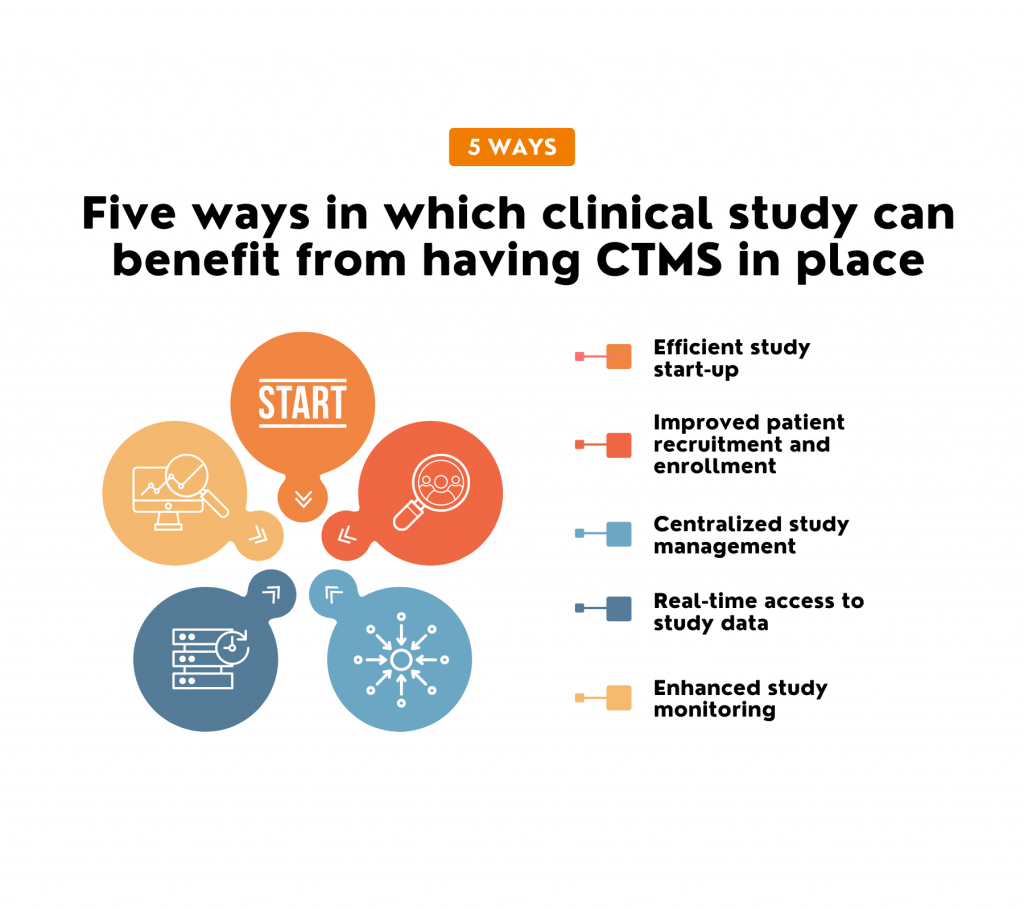
- Efficient study start-up: CTMS helps automate the study start-up process, including site selection, investigator contracts, and regulatory compliance. A study published in the Journal of Clinical and Translational Science found that implementing CTMS improved study start-up times, reducing the time required to initiate clinical trials by 50%.
- Improved patient recruitment and enrollment: CTMS helps to track patient recruitment and enrollment, identify recruitment issues, and facilitate communication between study staff and the project team. According to a survey by the Society for Clinical Research Sites, 75% of clinical research sites that use CTMS reported increased study enrollment. In comparison, 58% reported a reduction in screen failure rates.
- Centralized study management: CTMS provides a central hub for all study-related data, such as patient data, study protocol, and trial documentation.
- Real-time access to study data: CTMS provides real-time access to study data, which can help identify issues and make informed decisions during the study.
- Enchased study monitoring: CTMS is the best tool for monitoring study progress, including tracking the study milestones, site performance, and data quality.
And when it comes to Flex Databases, the further you get into digitalizing your processes, the more you earn. We can make the systems talk: patient enrollment from any of your EDC can be automatically transferred into CTMS, and monitoring reports from CTMS can go into eTMF. This is a humble example of data migration, saving time for multiple teams involved in a study.
Overall, CTMS can help improve the efficiency and accuracy of clinical trial management, reduce the risk of errors, and facilitate compliance with regulatory requirements.
Sounds like something you’d like to try? Send us a demo request to bd@flexdatabases.com to learn more!



