We’ve decided to compile an Ultimate Guide about Flex Databases CTMS and answer the biggest questions a person could have on the subject: from what is CTMS to what’s coming next.


We’ve decided to compile an Ultimate Guide about Flex Databases CTMS and answer the biggest questions a person could have on the subject: from what is CTMS to what’s coming next.

At the very beginning of 2022, Canadian CRO Nutrasource selected Flex Databases as an eClinical provider to cover the entire clinical trial lifecycle with a single solution.
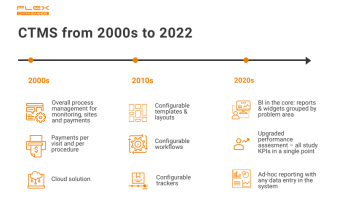
Clinical Trial Management System (CTMS) is one of the most popular and widely used eClinical software applications. It’s been on the market for decades, and from simple process management, the concept grew in many different directions. What happened to Flex Databases CTMS and where we are going now?
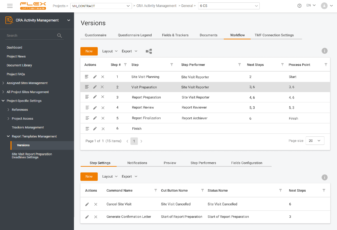
What if there is a way to do both – keep things the way they used to be yet go digital and get control over KPIs, overall transparency of the business and speed up processes?
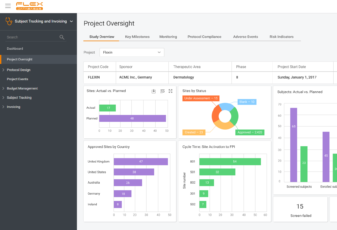
When we build a product, we base its core on the challenges our customers may face and how to solve them. We step in your shoes to see things from your point of view – what you need.

This summer ABX-CRO and Flex Databases signed a contract for a CTMS & eTMF implementation.

At the very beginning of 2021 Flex Databases and German FGK CRO signed a contract for CTMS and eTMF implementation.

At the very end of winter, Flex Databases signed a contract for CTMS and Pharmacovigilance module implementation with Italian Human & Digital CRO Exom Group.
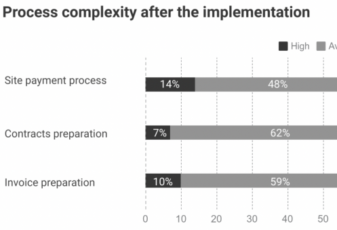
Most trial sites work with no more than a 3-month operating budget. With an estimated two-thirds of trial sites falling into this category, software companies have sought to close this gap in effective financial operations with process automation and payment schedule. So CROs, pharma companies, and biotech can finally get transparency in site payments.
Use Subject Tracking & Invoicing Module to track all patients’ data and manage invoicing online. Plan and schedule patient visits efficiently.
Bionical Emas is the only CRO to combine Clinical Development, Early Access Programs and Clinical Trial Supply, to deliver a unique, seamless approach supporting our clients to bring new medicines to patients faster.

United States-based Tranquil Clinical Research recently signed Flex Databases as a software provider for Clinical Trial Management System and Pharmacovigilance management solution.