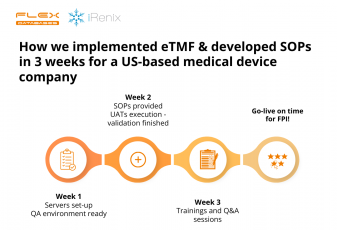
We are excited to share that Flex Databases has been named a Top Clinical Management Provider for 2024 by Life Science Review. This prestigious recognition highlights our strong reputation and the trust placed in us by both customers and industry peers, reflected in numerous nominations from Life Science Review subscribers. This award comes at a […]