Blog Flex Databases
📂 Last-minute TMF quality reviews cause stress, missed documents, and higher audit risks.With Flex Databases, you avoid the scramble – reviews are structured, transparent, and always under your control. With Flex Databases, you get:✅ Control over the process✅ Full transparency and traceability✅ Less stress before audits✅ Clear outcomes in one report 🎥 See how it […]
The FDA has just released its final guidance on Computer Software Assurance (CSA) for Production and Quality System Software. This marks a shift from traditional, document-heavy Computer System Validation (CSV) to a risk-based, critical thinking–driven approach. What does this mean? While this guidance is aimed at medical device production and quality systems, the principles perfectly […]
Ever uploaded a document to your Trial Master File only to realize it was already there?You’re not alone! Duplicates waste time, clutter your TMF, and create compliance risks. That’s why we built duplicate detection into Flex Databases eTMF. ✅ Real-time scanning as soon as you upload✅ Checks both file name and document content✅ Skip, replace, […]
Ever uploaded a document to your Trial Master File only to realize it was already there?You’re not alone! Duplicates waste time, clutter your TMF, and create compliance risks. That’s why we built duplicate detection into Flex Databases eTMF. ✅ Real-time scanning as soon as you upload✅ Checks both file name and document content✅ Skip, replace, […]
Applications for this position are now closed and no longer being reviewed. We are driven by our high mission to make an impact in the Life Sciences industry. We provide flexible e-Clinical software systems for Clinical Research Organizations (CROs) and pharmaceutical companies. We work globally with clients all over the world, with our own offices […]
When preparing a tender for a Clinical Trial Management System (CTMS) and an electronic Trial Master File (eTMF), CROs must choose solutions that meet sponsor expectations and improve operational efficiency while keeping studies inspection-ready. The right choice affects study delivery, compliance, and client satisfaction. Below are the key factors CROs should consider. Regulatory Compliance and […]
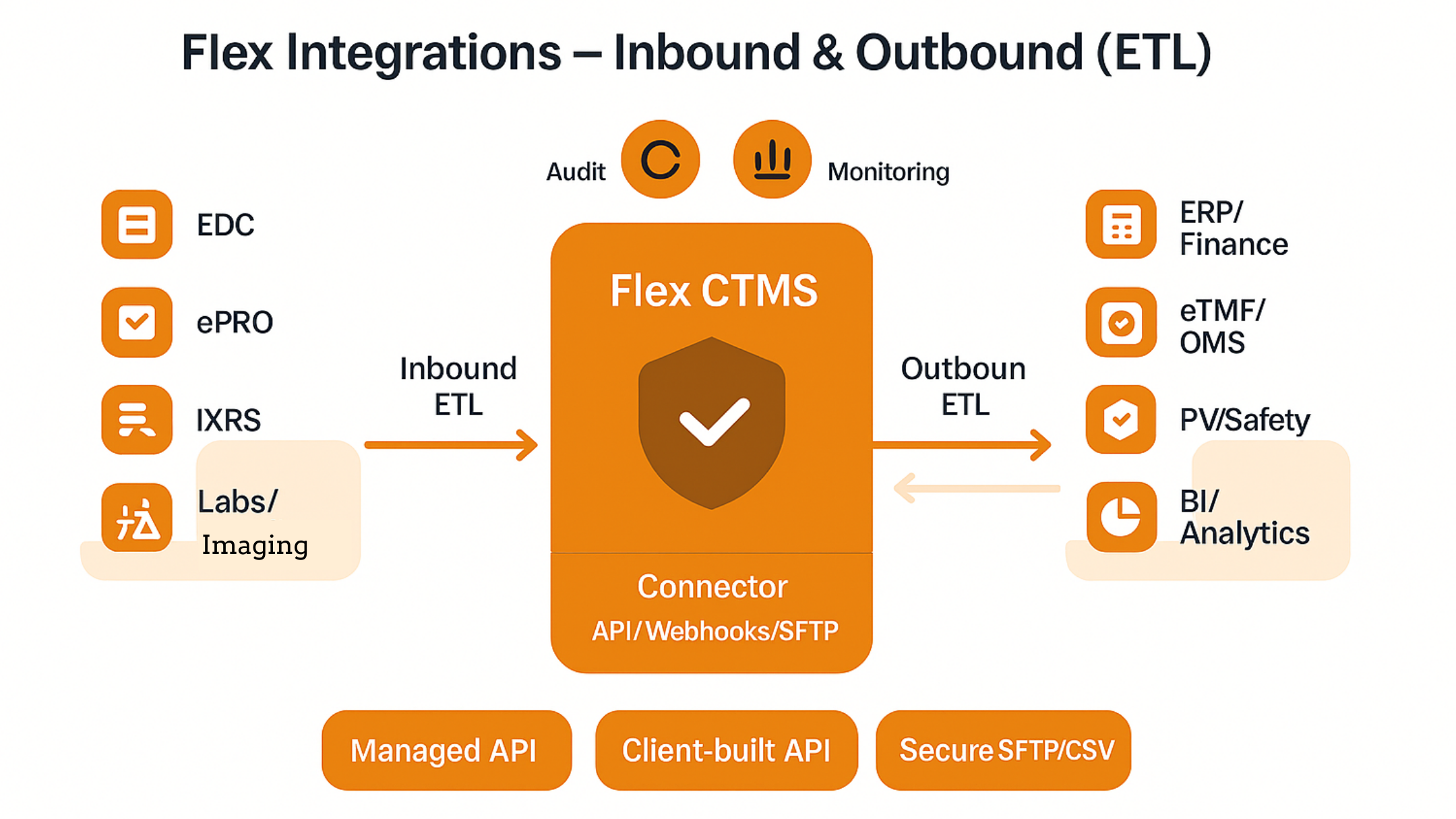
How Flex Databases connects your study ecosystem – reliably, securely, and at scale. Why integrations matter to sponsors and CROs Integration means that data entered once in a system of record is propagated automatically wherever it is needed. For clinical operations teams this translates into: Core integration patterns we see most often 1) EDC → […]
In July 2025, the European Commission adopted Commission Implementing Regulation (EU) 2025/1466, amending Regulation (EU) No 520/2012. The changes aim to strengthen pharmacovigilance, reduce unnecessary administrative burden, and align EU requirements with global best practices. Most provisions apply from 12 February 2026, with certain Eudravigilance updates taking effect in August 2025. Why the Changes Were […]
A Comparative Look at Europe vs. US Markets In clinical operations, time is money – but how much money can technology really save you? We analyzed the return on investment (ROI) for implementing electronic Trial Master File (eTMF) and Clinical Trial Management Systems (CTMS) based on real-world benchmarks. By modeling savings from reduced manual effort, […]
📍Remote 💼Full-Time Applications for this position are now closed and no longer being reviewed. We are driven by our high mission to make an impact in the Life Sciences industry. We provide flexible e-Clinical software systems for Clinical Research Organizations (CROs) and pharmaceutical companies. We work globally with clients all over the world, with our […]
What is Electronic Data Capture? Electronic Data Capture (EDC) is an electronic system applied to clinical trials for capturing, managing, and storing patient data. Instead of paper-based forms, investigators enter clinical trial information directly into a secure, web-based site. EDC systems validate data integrity, reduce errors, and automate the data collection process – accelerating clinical […]
At Flex Databases, we deliver a SaaS solution purpose-built for clinical trials – offering global accessibility with full transparency and control over where your data resides. Not a Public Cloud. A Dedicated SaaS Platform You Can Trust. Flex Databases is an EU-based company operating globally and trusted by life sciences organizations for secure, validated SaaS […]